 
In these equations R is the ‘Gas constant’ which is obtained by dividing the ‘Universal gas constant’ by molecular weight of a particular gas. 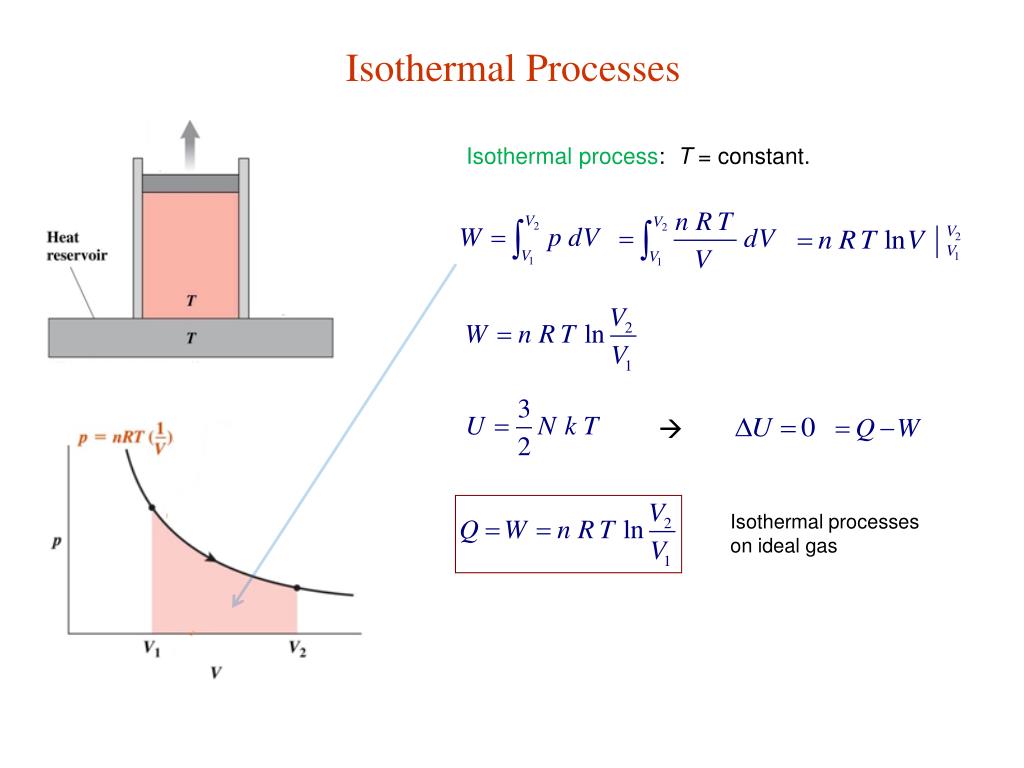
We get the head developed and power required for polytropic gas compression by replacing by n in the equations for adiabatic gas compression. If, n > the work done for polytropic compression and power required to run such a compressor is more than that required for and ideal frictionless adiabatic compressor. Note that when n is replaced by, this process becomes adiabatic. The actual compression processes are polytropic and typically described with the equation, Polytropic compression: Adiabatic (constant entropy) and isothermal (constant temperature) compression processes are ideal and theoretical in nature. Here subscripts 1 and 2 correspond to inlet and outlet states of the gas, V is the inlet volumetric flow of gas. This minimum possible power to compress a gas from P 1 to P 2 is given by following equation, The work done in this case and hence the power required to run this type of cooled compressors is the theoretically minimum possible limit. Isothermal compression: When the gas being compressed in a compressor is cooled with jacketed flow of a coolant, the process is an isothermal process. And as a consequence, the gas expands as the temperature does not remain constant resulting in a drop in compressor efficiency.This developed pressure head when multiplied by the inlet volumetric flowrate of the gas (V) gives shaft power required to drive an ideal adiabatic compressor, given by following equation, Isothermal compression is not possible in a true sense as the compression process happens so fast you will require infinite numbers of cold sources of heat sinks to keep the temperature constant. We are quite familiar with constant temperature and constant pressure isothermal phase transition like liquid to vapor. Actual compression processes are polytropic because the gas being compressed is not at constant entropy as in the adiabatic process, or at a constant temperature as in the isothermal processes. The work done by the surroundings in the irreversible process is numerically compared to the work performed in the reversible process. Real compression processes operate between adiabatic and isothermal compression. A closed system, on the other hand, can exchange only energy with its surroundings, not matter.Ģ. There are no changes in the control volume.Īn open system can exchange both energy and matter with its surroundings. This work, Vdp, is used for open flow systems like a turbine or a pump in which there is a “dp”, i.e. Today in our chemistry class we derived the pressure-volume work done on an ideal gas. ∫ VdP is the specific work for an open system. The gas will expand and perform work on the surroundings as long as appropriate heat is added to keep PV constant During isothermal expansion, both P and V change along an isotherm with a constant PV product. Calculate the final temperature, the work done, and the change in internal energy when 0.0400 moles of CO at 25.0 o C undergoes a reversible. 06 Calculate the work, heat, and change in internal energy when an ideal gas. Expert Answers: When an ideal gas is compressed adiabatically (Q0), work is done on it and its temperature increases in an adiabatic expansion, the gas does work and its. 
This can be equally considered as the maximum quantity of work. 
∫ PdV is the specific work for a closed system, as in a piston and cylinder. SKILLS YOU SHOULD HAVE MASTERED 1 Calculate the work done by a gas due to. W Work Done R Universal Gas Constant T Temperature V1, V2Initial and Final Volume, Work done in a reversible expansion of gas or work destroyed (or work applied to) in an reversible compression of an ideal gas can be calculated using the above chemistry calculator. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
